  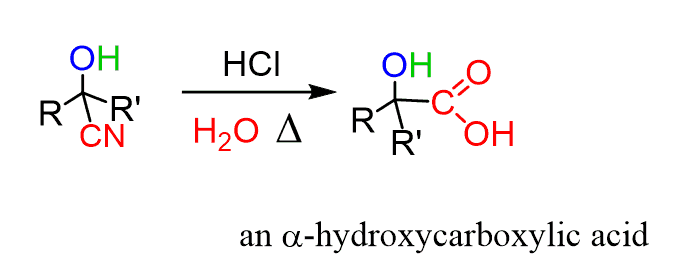
Product given with Strong oxidizing agent Now we are going to learn, which products are given by oxidation of different alcohol types. Oxidation of primary alcohol to an aldehyde. Pyridinium chlorochromate (PCC) is used as the mild oxidation agent +6 oxidation state of chromium in K 2CrO 4 is reduced to +3 Cr 3+ ion. Purple colour is changed to colourless or light pink. Strong oxidizing agents and their colour changes in alcohol oxidation Name You will learn more about these colour changes in next sections in this tutorial. Colour of oxidizing agent is changed when alcohol is Strong oxidizing agentsįollowing reagents are used as strong oxidizing agents in alcohol oxidation. Sometime, same alcohol will give different products for strong oxidizing agents and mild oxidizing agents. In alcohol oxidation, we use two types of oxidizing agents, strong and mild. When there are three alkyl group around the carbinol carbon, those alcohols are defined as primary alcohol. 
When there are two alkyl group around the carbinol carbon, those alcohols are defined as primary alcohol. When there are no alkyl groups or one alkyl group around the carbinol carbon, those alcohols are defined as primary alcohol. According to the type of alcohol, they response in different way for oxidizing agents. Primary, secondary, tertiary alcoholsĪccording to the number of alkyl groups around the carbinol carbon, alcohols are categorized as primary, secondary, tertiaryĪlcohols. According to the alcohol type, given product will vary. Oxidation, several oxidizing agents are used. Oxidation of primary, secondary, tertiary alcohols will give Oxidation of Primary, Secondary, Tertiary Alcohols to Aldehyde, Ketone, Carboxylic Acid 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
